Applications and Approvals
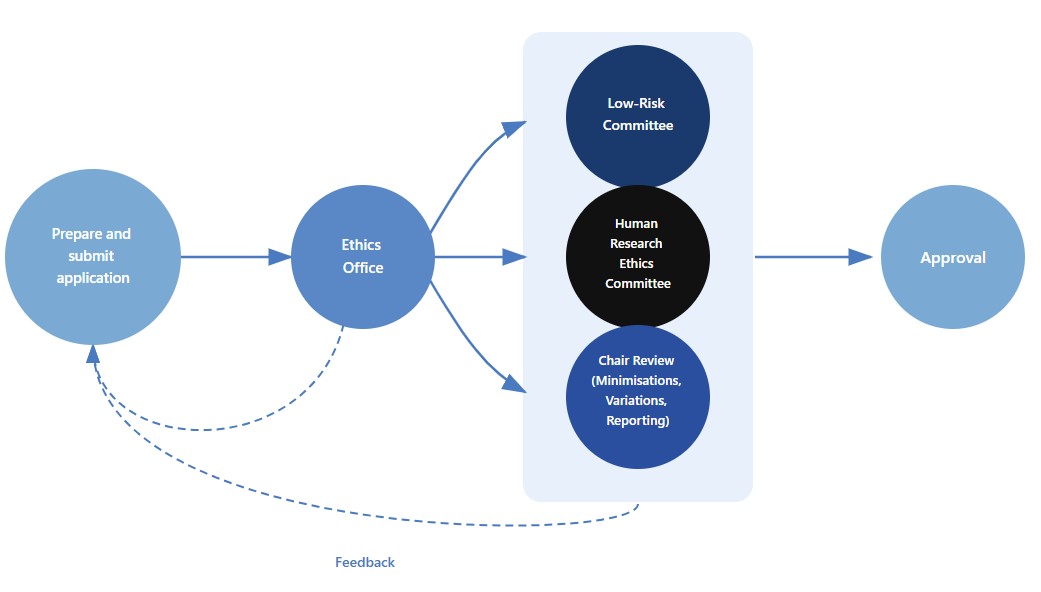
1. Prepare your application in the NHMRC’s HREA portal
- What it does: The Human Research Ethics Application (HREA) is designed to help you recognise potential risks in your project and show how you plan to manage them.
- When finished, download the HREA Output PDF. IMPORTANT NOTE: the HREA platform is not joined up to SCU platforms, clicking the 'Submit' button in the HREA does not send us an application.
- Guidance for the HREA is here
2. Assemble your supporting documents
- What they are: the accompanying materials the Committee needs to review your project (see the What to include tab, below, for the full list and templates). Save them as upload-ready files (PDF preferred).
3. Submit everything in IRMA (SCU’s ethics submission system)
- What it does: manages submissions and the review workflow.
In IRMA, create a new application and attach the HREA Output PDF and all required supporting documents, then submit. - Note: Only complete applications are sent for review.
- Guidance for IRMA is here. LInks to usage instructions may not be accessible for all users. Please use the interim link to IRMA navigation basics and coversheet instructions (sign in using your SCU credentials)
What happens next
Your submission is routed to the Human Research Ethics Committee or the Low Risk Committee, depending on the project’s level of risk.
From early 2026, new applications may require a faculty or college-level governance step before ethics review. This means your application must first be endorsed by your faculty or college to ensure it aligns with research priorities, resources, and compliance requirements before it proceeds to the formal ethics review process. More details to follow.
Before you compile your application
Confirm you've completed the SCU Online Ethics Training Module (now mandatory for all staff and students researchers). Evidence of completion must be included in your submission.
All applications must include the following
HREA form
Project description - full protocol OR brief low risk description (LRC)
Participant information sheet (PIS)
- Clinical Trials: see GCP informed consent requirements.
Consent form (where applicable)
- For anonymous online surveys, the PIS serves as the landing page with implied consent statement and a Yes/No checkbox – a separate signed consent form is not required.
Data collection tools
- Interview/focus group questions, survey instrument. observation protocols
- Surveys are typically hosted on Qualtrics; please provide a live survey link for review.
Recruitment materials
- Flyers, adverts, social media promotions phone/email scripts.
Research data management plan (RDMP). Now mandatory for all research projects, including student projects.
Full HREC review only
If applicable - depending on the research team and project
- Professional certifications (e.g. Working with Children check, GCP training)
- CVs for external (non-SCU) team members
- Evidence of peer review or governance approval
- HDR students: attach your Confirmation of Candidature report.
- AIATSIS training – evidence of completion (if working with Aboriginal and Torres Strait Islander peoples)
- Organisational support: correspondence with a gatekeeper or data custodians
Final notes
- SCU templates are designed to support efficient review; alternative formats are acceptable provided all required elements are addressed.
- Upload all documents as PDFs via IRMA.
- All participant-facing materials must show the SCU logo and a placeholder ethics number (e.g. 2026/XXXX).
- Please submit final versions and adopt appropriate document version controls.
- Applications cannot proceed to review if documents are missing or incomplete.
If your research involves human participants, you must obtain ethics approval before commencing. Human research includes any research with or about people, their data, or tissue. You need ethics approval to recruit, conduct research with participants, and you must maintain active ethics approval throughout analysis and until publication.
Apply as early as possible, but only once your research and documents are ready for review. Submissions must be complete (including all required attachments) before they can proceed.
Applications may be submitted at any time and will be processed on receipt. Early submission helps us triage your application and resolve any queries or clarifications out of session, so committee members can focus on the ethics issues at the meeting. Applications received after submission deadlines will be transferred to the following committee meeting.
Committee meeting and submission dates are found on the Ethics Committee page.
It is crucial to start preparing for ethics review as early as possible: treat it as integral to the project planning process. Plan for the review itself to take from two weeks to several months, depending on the complexity and risk level of your research, and the completeness of your application.
Check the committee meeting dates and submission deadlines and see Application review pathways below to ensure you allow enough time for feedback and revisions before starting your research.
See How long does the ethics review process take? on the FAQ page.
Tips for a speedy review include:
- Reviewers will react positively to thoughtful engagement with the ethical components of research design (see National Statement Chapter 3.1 for a ‘lifecycle of research’ approach)
- All necessary documentation must be final versions, and any public-facing materials must be SCU-branded.
- Attend to language – write for a lay audience, not your academic peers or your supervisor. Write your submission so reviewers can follow your decisions. Explain why you designed your project the way you did, and how you’ve taken ethical issues into account.
- Consider breaking complex research programs into their component phases – single methodology applications are easier to write and to review.
Regardless of who drafts the application, the Chief Investigator (CI) must sign off on it. As a rule, the CI should be based in a faculty, since all research at SCU must be hosted within one. Ethics submissions reflect upon the research team’s professionalism; it's important they are well-prepared. Students should seek guidance from their supervisors during the application process.
SCU staff or student credentials provide access to IRMA. If your SCU login details don’t work, instructions for creating an account are available in the Knowledge Base Notes.
SCU human ethics committees only accept submissions from SCU staff and students.
Non-faculty staff members seeking to lead research teams will be subject to approval from the DVCR before access to IRMA Human Ethics is granted. Your team may need to be reorganised to ensure faculty support. Adjunct researchers can not usually be approved as Chief Investigators.
According to the National Statement on Ethical Conduct in Human Research (2025):
Low risk human research describes research, including some types of clinical trials, in which the only foreseeable risk is no greater than discomfort. Discomfort is considered less serious than harm. It can involve physical or psychological impacts; for example, minor side effects of medication, discomfort related to non-invasive examinations or tests (such as measuring blood pressure), and mild anxiety associated with an interview. However, where a person’s reactions might exceed discomfort and become distress, this should be viewed as potential for harm. Any discomforts brought about by research should be effectively managed and minimised where possible. Low risk research is eligible for review by the SCU Low Risk Ethics Committee.
Higher risk human research is research in which the risk for participants or others is greater than discomfort. Research in this category carries risk of harm and therefore requires review by a Human Research Ethics Committee (HREC).
Where a person’s reactions might exceed discomfort and become distress, this should be viewed as harm rather than discomfort.
We encourage staff and students to engage with the Ethics Office, the HREC Chair, members of the University’s HREC or LRC for advice on risk level and the appropriate pathway for ethical assessment. Visit the Training and support page to watch The Ethics Edge: Episode 7 - Assessing Risk in Research. The final determination of the risk level of a research project is delegated to the HREC Chair, who consults HREC and LRC members to achieve consensus on the decision.
Helpful discussions about risk can be found in the National Statement on Ethical Conduct in Human Research:
- Chapter 2.1 – Risk and Benefit: This chapter assists researchers in identifying and articulating the level of risk involved in the research, strategies for risk minimisation, justification and management, and determining the appropriate level of ethical review in relation to the identified risks,
- Chapter 3.1 - Adopts a ‘lifecycle of research’ perspective, outlining the ethical challenges encountered at each stage of a project.
- Chapter 4.1 - This chapter discusses the recruitment of participants whose circumstances or experiences might mean that they are at increased risk from their involvement in research.
It is essential to engage with these chapters to ensure that your research aligns with the established ethical standards and that the risks are managed effectively.
The National Statement on Ethical Conduct in Human Research (2025) puts the spotlight on a thoughtful, project-by-project risk assessment. The key question is whether your research could expose its participants to risks beyond their everyday experience of discomfort and recognising when specific research contexts or issues to do with the way the project is designed could heighten the risks to the people in your study. When it does, an HREC review is required.
Research ethics at SCU is moving beyond protectionism towards an inclusive, proportionate, and transparent approach. The emphasis is on managing risk appropriately by enabling participation, adopting a strength-based and context-sensitive process, and ensuring ethics governance supports inclusion rather than exclusion.
|
When the National Statement Still Requires HREC Review While the participant categories listed in the 2023 version are no longer automatic triggers, the National Statement (2025) still directs some types of research to HREC review: |
|---|
|
Participant populations where combinations of risk increase the likelihood of harm Research contexts likely to increase the risk of harm Waivers of consent and challenges to consent in certain higher risk research contexts |
This tool helps users identify whether ethics review is required and, if so, whether review by the low‑risk committee or HREC is most appropriate. Final determinations rest with the relevant review body, and advice from the Ethics Office may be required in complex cases.
Low Risk Review
The LRC meets fortnightly to review applications submitted in accordance with the schedule. The review response time frame is 15 working days from the submission deadline.
If you believe LRC review is appropriate for your project, you will be asked to include a brief (1-2 paras) rationale at submission. Refer to chapters 2.1 (Risk and Benefit) and 4.1 (Recruitment and involvement of research participants who may experience increased risk) of the National Statement.
Note that the LRC Chair may refer projects involving sensitive or controversial topics to the HREC for review.
High Risk Review
The HREC meets monthly to review applications submitted in accordance with the schedule. The review response time frame is 20 working days from the submission deadline.
Minimisation of Duplication
The National Statement (Chapter 5.5) encourages the minimisation of any unnecessary duplication of ethics review both within and across research sectors. This extends to research that will be conducted in more than one Australian jurisdiction or across international boundaries. If you already have ethics approval for your project from another HREC, IRB or equivalent ethics review process, SCU staff and students are encouraged to register their approval with us by submitting a Minimisation via IRMA.
Provide the ethics approval from the host institution and all documents submitted and approved by them, including any approved changes of protocol.
Alternatively, if you are transferring to SCU from another university and you want SCU to oversee your project, ensure the original HREC agrees to SCU assuming responsibility and provides correspondence to this effect, then submit application and approval documents from the external HREC but choose ‘New Application V4’ and specify in the coversheet that you are requesting a TRANSFER.
Collaborative research projects conducted under the auspices of SCU should display university branding on outward-facing materials to ensure visibility and transparency. If branding is not appropriate for research-related reasons, please explain this in your submission. We also expect SCU researchers to be named with their affiliation and role in the project, to reflect appropriate recognition and responsibility.
Amendments to Minimisations
Please submit documentation for any amendments to Minimisations that have already been approved elsewhere using a Change of Protocol form in IRMA and clearly label it as a change to a Minimisation.
This allows us to link the update to the original application without re reviewing the approved conditions.
Minimisation applications are reviewed and approved by the Human Research Ethics Chair.
See also the FAQ: How do I request a Minimisation of Duplication of ethics review
Exemption
The HREC Chair may choose to grant exemption from ethics review for low risk research that meets one or more of the conditions in section 5.1.17 of the National Statement.
To apply for an exemption, researchers must review sections 5.1.15- 5.1.18 of the National Statement and email the following to the Ethics Office:
- A 2-page (max) description of the activity
- A justification letter addressing the specific criterion of the National Statement in section 5.1.17 (a)-(d), that applies, and confirming that the activity is not disqualified under section 5.1.16.
- Any relevant project documents (eg participant documentation or gatekeeper correspondence).
The HREC Chair will assess the application with the template, Exemption from Review Assessment , and issue a letter of approval or rejection within 10 business days.
The Ethics Office will keep records of research exempted from ethics review. Researchers must do the same.
Researchers must keep an auditable record of any research they are undertaking that is exempted from ethics review in accordance with NS 5.1.15. This is required to ensure that there is a record of the research where no review has been conducted.
After the review, there are five possible review outcomes:
- Approve
- Approvable with minor amendments back to Chair (no submission deadline)
- Approvable with minor amendments back to Chair and Spokespeople (no submission deadline)
- Revise and resubmit to submit to the relevant committee (submission deadline applies)
- Desk reject (requires a new submission to be made)
- Rejection
LRC review
The Chief Investigator will receive an outcome notification via email two weeks after the submission closing deadline
HREC review
The Chief Investigator will be notified of the HREC review outcome via email within two weeks of the committee meeting.
The HREC and LRC and subcommittees expect any issues to be addressed within six weeks to prevent applications from lapsing. Researchers should plan accordingly to allow sufficient time for the ethics review process.
The Chief Investigator is responsible for obtaining ethics approval for all changes of protocol before they are implemented. Failure to adhere to the approved protocol constitutes a breach of ethics and may be research misconduct. It can jeopardise the researchers' legal indemnity, publication rights, and future funding opportunities.
Apply for a Change of Protocol (COP) using IRMA. (Guidance here)
Changes of protocol may include, but are not limited to:
- Adjustments to the research protocol
- Modifications to recruitment strategies
- Updates to participant information sheets and consent forms.
- Revisions to letters, advertisements, questionnaires, or other documents
- Alterations to the research team composition
When public documents such as participant information statements, consent forms, advertisements, and questionnaires are modified, amended documentation should be included with the change of protocol request. Please provide both tracked and clean copies of any revised documentation as PDF files. This ensures efficiency, transparency and adherence to ethical standards throughout the research process.
Ensure that you provide a rationale for the changes requested.
Is this a change of protocol, or a new project?
A COP is for changes that maintain substantive continuity with the project as originally approved. It is not a mechanism for extending a completed project or initiating what is, in substance, a new research endeavour.
Use a COP when changes...
- Refine how an ongoing project is conducted
- Involve participant groups anticipated in the original protocol
- Adjust recruitment within the originally described population.
Likely a new project when...
- The project has been completed and you wish to reinstate it
- The proposed cohort is materially different from, and was not envisaged in the original protocol
- The research questions, aims, design, or risk profile have changed substantially (that is, the proposed work would not have been recognisably described by the original application).
New projects require governance review in addition to ethics approval. If you are unsure whether your change requires a new application, contact the Ethics Office before submitting to avoid delays.
Annual progress reports
Research teams will be emailed reminders to submit the following reports via IRMA.
You must submit an annual report in IRMA, in accordance with section 5.4.8 of the National Statement, to confirm that the research remains consistent with its approved ethical framework. This includes updates on progress, data security, and adherence to approved protocols and conditions.
Reminders for overdue reports are issued at 7 and 30 days past deadline.
Failure to respond within 60 days will result in a withdrawal of ethics approval.
Renewal requests
You will need to apply for project renewal at least one month before the current approval expires, including reasons and a need for extension in compliance with Section 5.4.10 of the National Statement.
If changes to the project are planned that alter its scope, amplify participant risks or necessitate additional risk mitigation, a Change of Protocol should accompany the renewal application.
Each renewal is evaluated individually, and if approved, the team is officially informed of an additional three-year ethical endorsement.
Final report
You should submit a final report when:
- your research activities with participants have concluded,
- you no longer plan to contact participants for data collection or verification, and
- you have either produced the final written output (thesis, report or publication), or decided not to publish.
Monitoring/Audit
Maintain an auditable record of your entire research project. From time to time, research projects at SCU may be selected for an ethics audit. Monitoring helps ensure projects continue to meet approved ethical standards and protect participants. If your project is audited, you will be asked to provide consent documentation, recruitment materials, data protocols, and progress reports. Audits can be routine, random, or triggered by reports or concerns. Researchers are expected to cooperate fully, respond to requests promptly, and address any corrective actions identified. Keep clear records of every stage of your project – from planning and approval through to completion and dissemination, so they can be audited if required.
Southern Cross University takes all complaints seriously, whether they relate to the conduct of research or the operations of its ethics committees. All complaints are handled with sensitivity and confidentiality and may be submitted by anyone: research participants, researchers, staff, students, or members of the public.
Complaints about the ethical conduct of research
If you have concerns about how a research project is being carried out, you can raise them with the Ethics Office.
Complaints about the conduct of the Ethics Committees
If you have concerns about how an Ethics Committee is operating, you can raise the issue with the Office of the Senior Deputy Vice-Chancellor, if you are uncomfortable raising it directly with the Ethics Office.
Research Integrity Complaints
If you suspect a potential breach of the Australian Code for the Responsible Conduct of Research, 2018, you can make a complaint through the Research Integrity Database. SCU staff and students are encouraged to first seek advice from a Research Integrity Advisor in your Faculty or College. Further information on Research Integrity is on the Governance and integrity page.
Adverse events are any unfavourable occurrences, outcomes or responses that affect your research participants, yourself as a researcher, or the reputation of your institution.
What is a serious adverse event?
A serious adverse event is one that results in significant health impacts, such as death, life-threatening situations, hospitalisation, or persistent/significant disability.
Reporting requirements:
- All adverse events – serious or not, and even if resolved – must be reported to the Ethics Office using the Adverse Event Form in IRMA.
- Serious adverse events: Report within 72 hours of the event. If you’re unsure about the severity, call or email the Ethics Office immediately for advice.
- Other adverse events: Report promptly (within 7 days of becoming aware of the event).
- Minor/manageable events: Report through the Adverse Event Form in IRMA. If expected and low-impact, these may be summarised in annual or final reports, unless part of a broader pattern.
- Protocol deviations: Any changes or departures from the approved protocol that may affect participant welfare or ethical integrity must be reported immediately.
Clinical trials of unapproved goods:
Follow the TGA’s expedited reporting framework Report a problem or side effect. Therapeutic Goods Administration (TGA) and the NHMRC’s Safety monitoring and reporting in clinical trials involving therapeutic goods.
Encouraging participant reporting:
Researchers should encourage participants to report any concerns or incidents directly to them or to the Ethics Office.
Further guidance:
For more details on clinical trial reporting, refer to the ICH Guideline for Good Clinical Practice.
An ethics breach is any violation of the approved ethics protocol that impacts the acceptability of your research. Ensure you report all breaches to the Ethics Office promptly via IRMA.
Remember that breaches can include conducting unauthorised research, straying from approved protocols, and failing to comply with national ethical research guidelines. These may occur as isolated incidents or repeatedly.
Should an ethics breach be identified, the HREC Chair has the authority to escalate the issue to the Research Integrity Office. It could lead to discussions, investigations, or resolutions, particularly if it involves infractions related to the Australian Code for the Conduct of Responsible Research.
Consider revising your protocol as this can often remedy ethical breaches.
Once an ethics violation is resolved, make sure it is reported back to, and acknowledged by, the Ethics Office.

/prod01/channel_8/media/scu-dep/current-students/images/Coffs-harbour_student-group_20220616_33-147kb.jpg)
/prod01/channel_8/media/scu-dep/current-students/services/counselling/images/RS21533_English-College-Student_20191210_DSC_6961-117kb.jpg)
/prod01/channel_8/media/scu-dep/study/scholarships/images/STEPHANIE-PORTO-108-2-169kb.jpg)
/prod01/channel_8/media/scu-dep/study/arts-and-humanities/images/RS20958_Chin-Yung-Pang-Andy_20190309__79I5562-960X540.jpg)
/prod01/channel_8/media/scu-dep/experience/images/SCU-INTNL-STUDY-GUIDE-280422-256-72kb.jpg)
/prod01/channel_8/media/dep-site-assets/component-library/screenshots/online-1X1.jpg)